  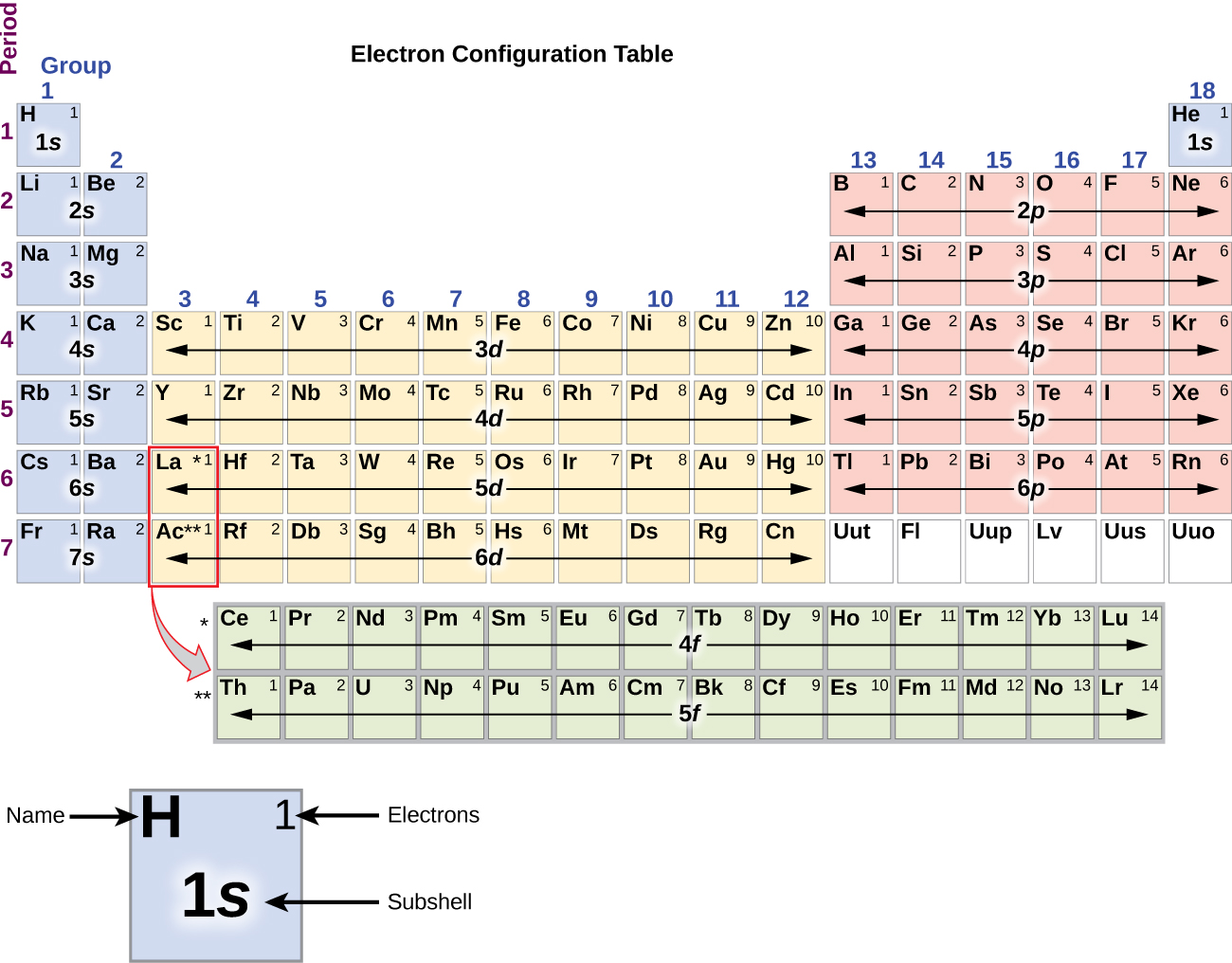
Number of atoms (or add up each atom individually).įind the molecular mass of MgO. If there are multiple atoms of an element, multiple by the stands for atomic mass units-Īdd up the masses of the individual atoms. Is in is its number of valence electrons.įind the valence electrons for the following elements: Using the Periodic Table The last digit of the Group (column) an element 
All other elements will lose, gain, or share to reach 8 electrons. “If I 8, I full.” Only Group 18A have a full octet Octet Rule – Atom are more stable that have a full shell of electrons. Valence electrons are theĮlectrons that are involved in chemical bonding. Atoms share electrons and form covalent bonds to satisfy the octet rule. 4.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. The atoms in group 6A make two covalent bonds. (column) has the same number of valence electrons. Covalent bonds are formed by two atoms sharing electrons. Atoms in a period have the same number of electron shells. An element period is a horizontal row on the periodic table. Valence Electrons are the outermost electrons in an atom. Atoms in a group share the same number of valence electrons. Period 2 is row 2 Group 16A is Column 16A. What element is in Period 2 and Group 16A? Thinking like the game “Battleship”, you can Hydrogen is in the same group as Lithium. On the periodic table groups =Ĭolumns (up and down). Valence electrons varies 1 H 3 Li 19 K 11 Na 4 Be 20 Ca 12 Mg 5 B 31 Ga 13 Al 6 C 32 Ge 14 Si 7 N 33 As 15 P 8 O 34 Se 16 S 9 F 35 Br 17 Cl 2 He 10 Ne 36 Kr 18 Ar 1A 18A 2A 13A 14A 15A 16A 17A 1 2 3 4Įlements that have the same number of outermost electrons have similar Stephen Murray, 2003 Periods and Groups Valence Electrons Masses 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
